Photoactive Materials for Photonics and Healthcare
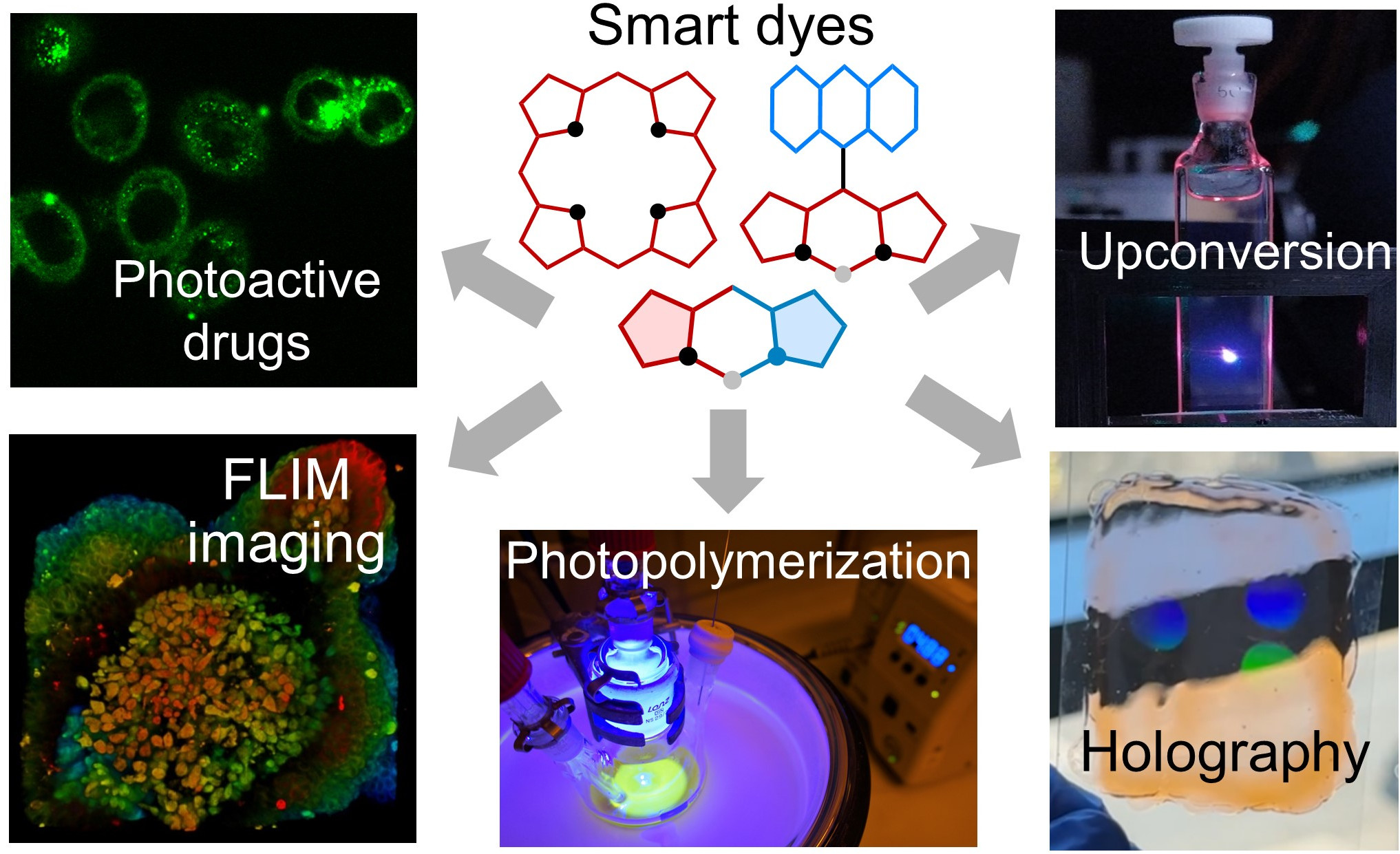
A core activity of our group is translating functional dyes into photoactive materials - formulations and platforms where light triggers controlled transformations and measurable optical outputs. Together with synthesis and photophysical studies, this translation step connects molecular design to applications spanning, photonics, advanced manufacturing, imaging, sensing, and phototherapy.
Photopolymerization and Holography
A major translation route is light-driven polymer formation, where photoexcited dyes generate reactive intermediates that initiate curing under visible light. In our work, chromophores engineered for efficient triplet formation and charge-transfer reactivity function as visible-light-activatable photoinitiators in multicomponent formulations, enabling rapid photopolymerisation under mild conditions and providing a practical handle to tune curing kinetics and material performance.
Materials for Triplet–Triplet Annihilation Upconversion (TTA-UC)
In triplet–triplet annihilation upconversion (TTA-UC), two low-energy photons are effectively combined into one higher-energy photon, enabling visible emission under long-wavelength excitation. This provides a practical route to harvest red/NIR light and convert it into a brighter, higher-energy optical output for light-management technologies and photonic applications.
Our work on TTA-UC materials focuses on three complementary directions:
- NIR-absorbing upconversion materials. Sensitizer platforms that extend excitation into the red/NIR, enabling conversion of ~600–800 nm light into visible emission.
- Oxygen-resistant systems. Formulations with integrated oxygen scavengers that suppress triplet quenching and protect active components from oxidative degradation, extending device lifetimes.
- Dual-performance dyes. Systems where a single dye can act as either the triplet sensitizer or the emitter component of the TTA-UC process depending on the medium, enabling a switchable functional role through formulation and environment.
The video below shows an example of a TTA-UC device converting 805 nm NIR laser excitation into visible emission.
Palladium and platinum complexes of tetraaryltetraanthraporphyrins developed by us enable the energetically-conjoined TTA-UC process with rubrene as an emitter, when excited with NIR light (800 nm), at ultralow light intensity of <100 mWcm-2.
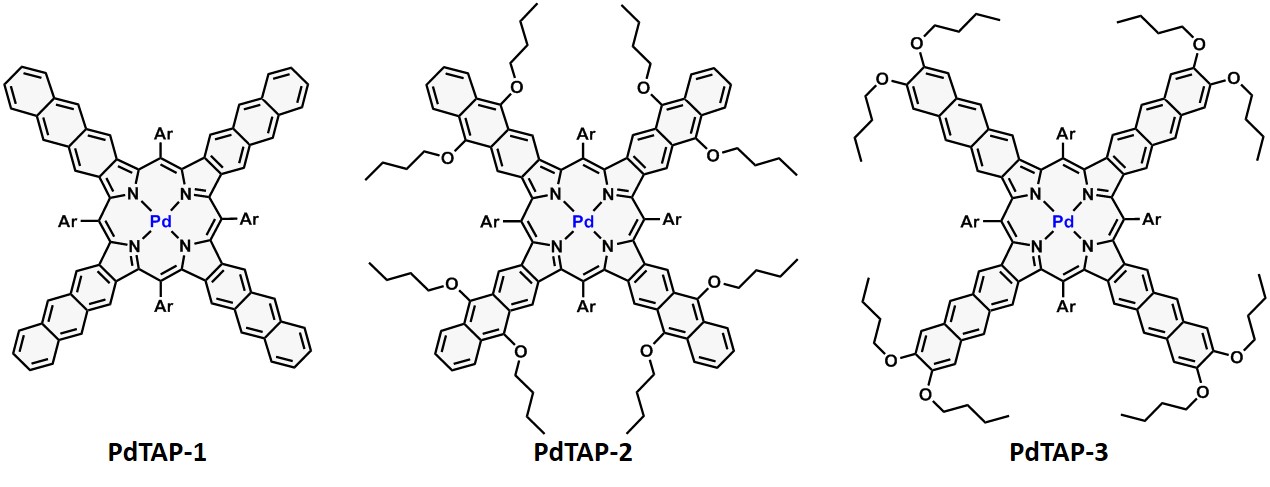 Structures of palladium(II) tetraanthraporphyrin sensitizers. |
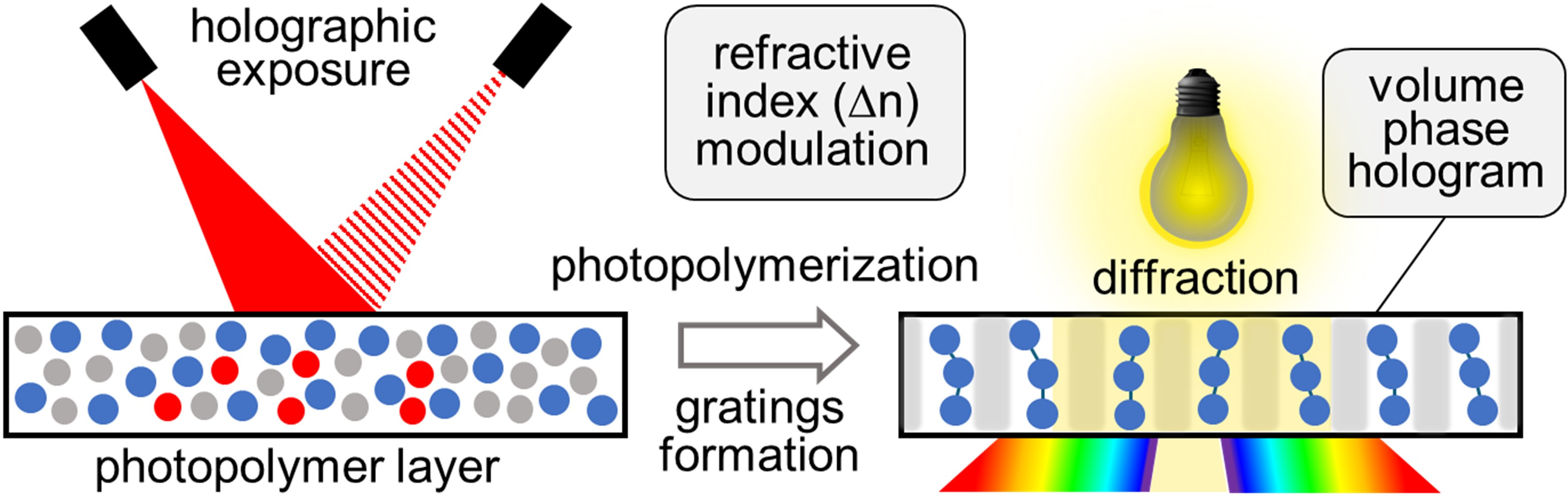
In these devices, tetraanthraporphyrin (TAP) sensitizers transfer triplet energy (TTET) to a single emitter species (rubrene). A mixture of sensitizers with different absorption maxima was used to broaden the overall excitation profile for upconversion. The optical properties of TAP enable upconversion across the 720–840 nm region. Broadband excitation in this range with simulated moderately concentrated sunlight (integral intensity ~ 50 mWcm-2) produced efficient upconversion emission (>4% quantum yield).
To address oxygen sensitivity, functioning UC devices were prepared and sealed under inert conditions using a specially designed liquid-matrix system that provides active protection against oxygen. Under these conditions, the devices operated stably for several months without noticeable performance degradation, even when placed under water.
 Images of a UC device based on PdTAP sensitizers and rubrene emitter, irradiated with an 806 nm diode laser and emitting yellow light (left). The same device placed under water (right). |
Fluorescent Probes for Sensing and Bioimaging
A complementary direction is information readout: dyes designed so that their emission intensity and/or lifetime reports on the local microenvironment. Fluorescence microscopy visualises the spatial distribution of emissive probes in cells and tissues, while fluorescence lifetime imaging microscopy (FLIM) maps the emission lifetime, which can provide concentration-independent contrast and report on local environment and excited-state pathways.
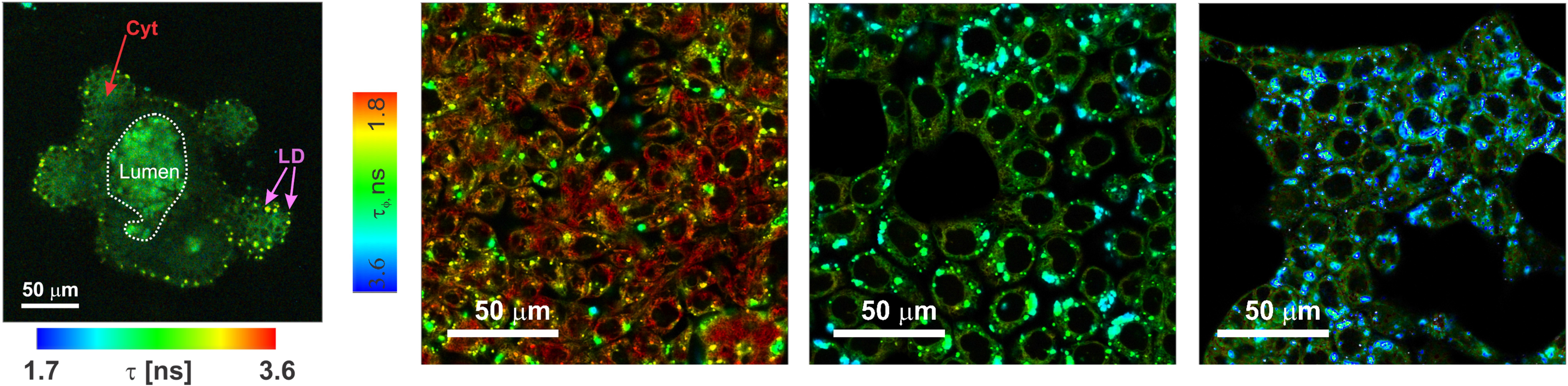 Fluorescence lifetime imaging microscopy (FLIM) of a monolayer of colon HCT116 cancer cells. The colour-coded lifetime map provides concentration-independent contrast and reports on microenvironmental and excited-state variations across the cell population. |
Singlet oxygen is an electronically excited state of molecular oxygen that is less stable than ground-state (triplet) oxygen. It is typically generated via energy transfer from the triplet excited state of a photosensitizer to the oxygen molecule. Singlet oxygen is highly reactive and is used, for example, to destroy cancer cells in photodynamic therapy.
Intracellular singlet oxygen is thought to be involved in physiological and pathological processes such as signaling, immune response, and induction of gene expression. However, detailed molecular understanding of its roles has been limited by the lack of suitable detection and imaging techniques. Fluorescein-based Singlet Oxygen Sensor Green (SOSG®), the only available commercial probe, was shown to be cell-impermeable, susceptible to photodecomposition, and to generate ¹O₂ under light irradiation.
Probes for singlet oxygen are in demand across immunology, oxidative stress, pharmacology and cancer research. Currently, we investigate new BODIPY-based probes to monitor singlet oxygen generation. Reaction with ¹O₂ produces a strongly fluorescent product, enabling visualisation of singlet oxygen generation in living systems; such fluorogenic behaviour has been demonstrated in human breast cancer cells.
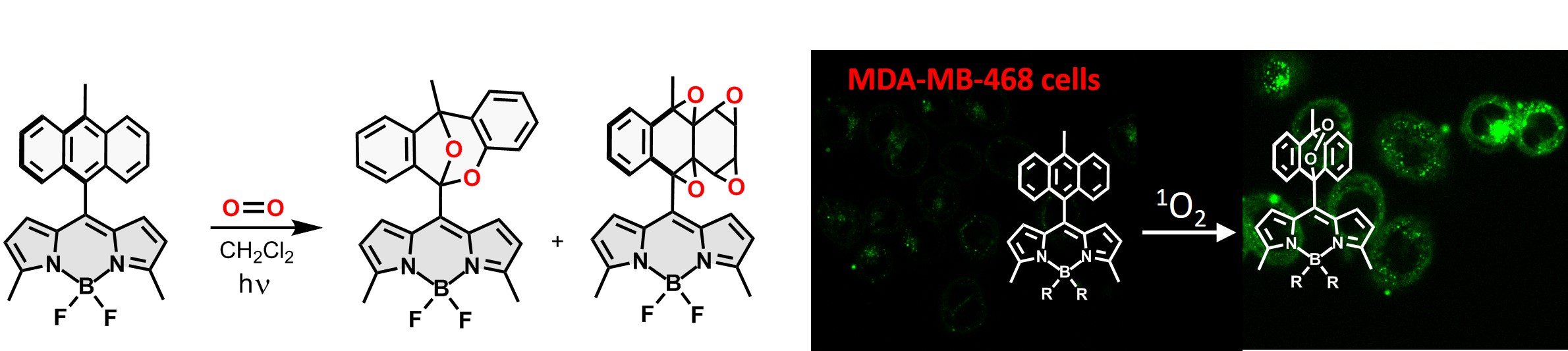 Reactions of BODIPY probe with singlet oxygen leading to fluorogenic response in living cells. |
Materials for Photomedicine and Theranostics
Triplet-state chemistry enables controlled generation of reactive oxygen species and related outputs relevant to biomedicine. Representative examples include porphyrin-based platforms capable of generating, storing, and thermally releasing singlet oxygen, providing a route to decouple light exposure from downstream oxidative chemistry.
We develop molecular systems capable of generating, binding, and releasing singlet oxygen molecules. An example—tetrabenzoporphyrin with appended anthracene groups—is shown below. Upon irradiation (or exposure to daylight) in air-saturated solution, this compound forms a corresponding endoperoxide, a product of a [4+2] cycloaddition reaction between singlet oxygen and anthracene. Release of the bound oxygen can be triggered by heating.
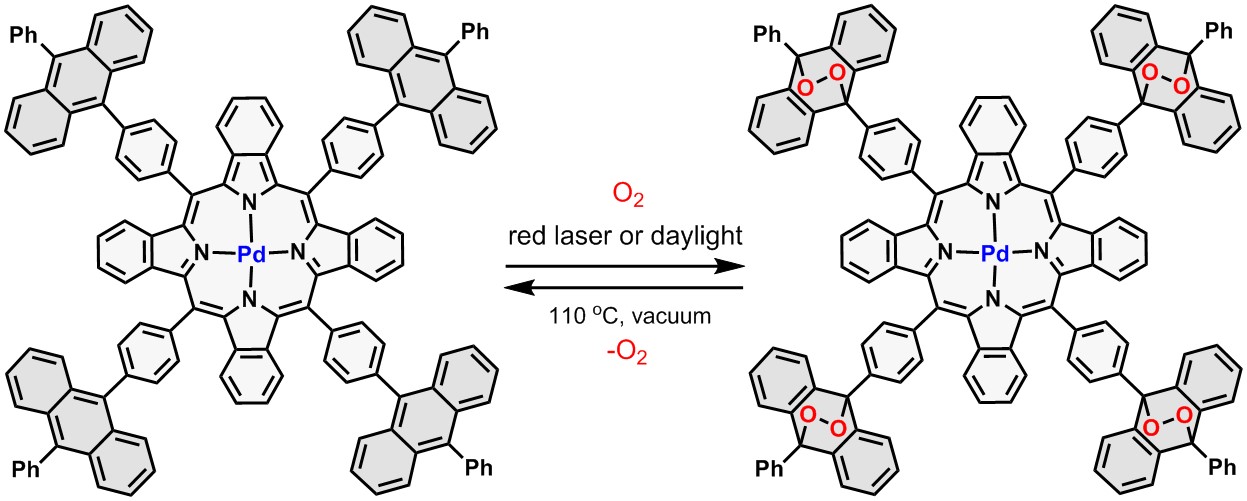 Binding and release of singlet oxygen by palladium(II) tetrabenzoporphyrin. |
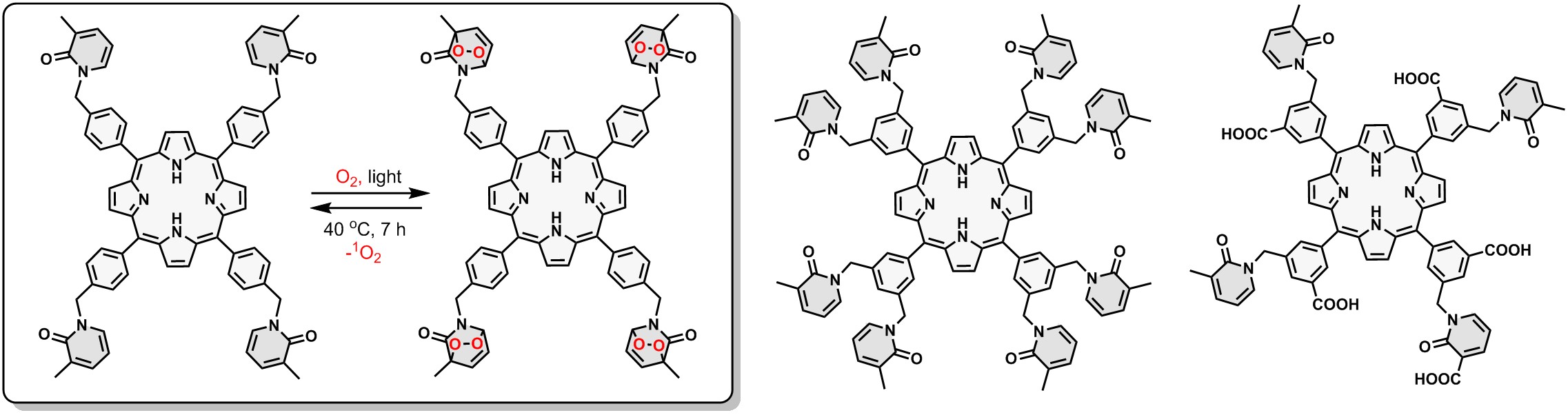 Pyridone appended porphyrins - controlled singlet oxygen release sensitizers for photodynamic therapy. |

This research is supported by Research Ireland (Taighde Éireann).
